
In the full quantum mechanics, one may encounter several "quantization" facts with the quantum proportional to $h$ or $\hbar$ or $\hbar/2$. And it doesn't make too much sense to think about the origin of the conditions in the Bohr model because the Bohr model is fundamentally not the right theory as we know today. One must separate the explanations in the Bohr model from those in the proper quantum mechanics they're inequivalent because the models are inequivalent, too. In quantum mechanics, similar conditions hold but for slightly different reasons and the quantization of the angular momentum allows half-integral values, too: $J=n\hbar/2$ where $n$ is integer according to quantum mechanics. The orbit encircles an area in the phase space and the area should be quantized.īy a coincidence, this is also pretty much equivalent to the quantization of the angular momentum, $L=n\hbar$. In this form, it is analogous to the statement in the full quantum mechanics – that replaced the Bohr model in the mid 1920 – that the phase space (space parameterized by the positions $q$ and momenta $p$) is composed of cells whose area (or volume) is equal to multiples of $h=2\pi\hbar$ (or powers of $h$, if there are many coordinates). 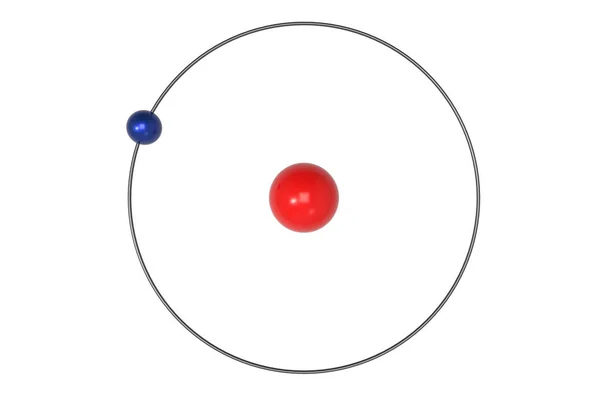
Where the integral of $p\,dq$ goes over one orbital period. The integrality conditions of the Bohr model were ad hoc – chosen so that the energy levels as seen in the Hydrogen absorption/emission spectra could be matched – but the most justified starting point to derive them was the Sommerfeld-Wilson quantization condition 1787 nm difference).The Bohr model wasn't the right theory of all of atomic physics but it described the levels of the Hydrogen atom correctly, due to a mathematical coincidence related to this solvable mathematical problem in the full quantum mechanics. The red line of deuterium is measurably different at 656.1065 (. A rough classification of some ofthe types of radiation by wavelength is: Radiation of all the types in the electromagnetic spectrum can comefrom the atoms of different elements. The three prominent hydrogen lines are shown at the right of the image through a 600 lines/mm diffraction grating.Īn approximate classification of spectral colors: The colors cannot be expected to be accurate because of differences in display devices.Īt left is a hydrogen spectral tube excited by a 5000 volt transformer. It was viewed through a diffraction grating with 600 lines/mm. This spectrum was produced by exciting a glass tube of hydrogen gas with about 5000 volts from a transformer. Electron level calculationįurther splitting of hydrogen energy levels And even the 1s ground state is split by the interaction of electron spin and nuclear spin in what is called hyperfine structure. The 2s and 2p states are found to differ a small amount in what is called the Lamb shift. The 2p level is split into a pair of lines by the spin-orbit effect. If you look at the hydrogen energy levels at extremely high resolution, you do find evidence of some other small effects on the energy. The energy levels agree with the earlier Bohr model, and agree with experiment within a small fraction of an electron volt. The basic structure of the hydrogen energy levels can be calculated from the Schrodinger equation. If the radial probabilities for the states are used to make sure you understand the distributions of the probability, then the Bohr picture can be superimposed on that as a kind of conceptual skeleton. This Bohr model picture of the orbits has some usefulness for visualization so long as it is realized that the "orbits" and the "orbit radius" just represent the most probable values of a considerable range of values. Common pictures are those of a shell structure with each main shell associated with a value of the principal quantum number n. 
Hydrogen Energy LevelsThe basic hydrogen energy level structure is in agreement with the Bohr model. But for heavier elements the movement of the nucleus can be neglected. So the reduced mass of the electron is needed. The reason for the variation of R is that for hydrogen the mass of the orbiting electron is not negligible compared to the proton at the high accuracy at which spectral measurement is done. This is often expressed in terms of the inverse wavelength or "wave number" as follows: 
Hydrogen energies and spectrum Electron TransitionsThe Bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
